Need Help?
Ask an expert
1. How to collect Salivary C-Reactive Protein
APPROVED SALIVARY C-REACTIVE PROTEIN COLLECTION METHODS
Salivary C-Reactive Protein Collection Protocol
Collection volume, general considerations, and basic guidelines to maximize salivary C-Reactive Protein sample integrity. Use this analyte-specific collection protocol to plan you collection methodology and sampling schemes.

2. How to Assay for Salivary C-Reactive Protein
Send Saliva Samples to Salimetrics
Add to StudyEasy and accurate results from the Salimetrics CoreLab+ Laboratory.
Order Code5417
Salivary C-Reactive Protein ELISA Kit
Add to Study
Salimetrics Assay #1-2102 (Generation II)
The Salimetrics Salivary C-Reactive Protein (CRP) Enzyme Immunoassay Kit (Generation II) was specifically designed to standardize the detection of CRP in saliva samples for research and biomedical laboratories. Using a small sample volume, this assay kit has an extended range that spans the expected CRP levels found in human saliva. The average inter- and intra-assay precision coefficients of variation are low with no deleterious matrix effects often found in saliva, which are characterized through dilution- and spike-recovery validation procedures. This CRP assay kit has also been formatted to minimize cross reactivity for related cytokines. CRP levels in humans are normally quite low, but they increase several hundred fold during the acute-phase response. Studies have shown that CRP levels can be linked to the incidence of heart attacks and strokes, and can be used to monitor general cardiovascular health and as a predictor of future coronary events. Numerous recent studies investigating serum CRP and its relationship to other diseases have included hypertension, diabetes, cancer, and autoimmune disorders. These studies also suggests possible links between oral health and chronic infection, inflammation, and heart disease. Read More...| Assay Protocol |
|---|
| Rev. 05.31.24
|
| Specifications | |
|---|---|
| Catalog#: | 1-2102 |
| Regulatory Status: | RUO |
| Format: | 96-well plate |
| Assay Time: | ~ 5 hrs |
| Sample Volume/Test: | 150 µL of saliva, 100 µL of x2 dilution |
| Sensitivity: | 1.79 pg/mL |
| Assay Range: | 25 pg/mL - 1600 pg/mL |
| Storage Requirements: | 2-8°C |
| Tests Per Kit | |
|---|---|
| Singlet: | 76 |
| Duplicate: | 38 |
| Target Analyte |
|---|
Technical Documentation
Assay Kit Overview
Intended Use
The Salimetrics® Salivary C-Reactive Protein ELISA Kit (Generation II) is an enzyme-linked immunoassay specifically designed and validated for the quantitative measurement of human CRP in oral fluid. It is not intended for diagnostic use. This assay kit was optimized for human salivary research and has not been validated for other human sample types, such as serum or plasma or samples from other species.
Introduction
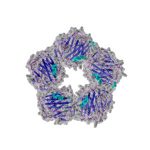
C-Reactive Protein (CRP) circulates in the blood plasma as a stable homopentamer and a prominent member of the acute-phase inflammatory proteins. CRP functions mainly in innate immune defense and levels increase in response to inflammation, infection, tissue damage, necrosis, malignancy and allergic reactions. CRP has diagnostic value as a marker of systemic inflammation in the body and as an independent risk factor for cardiovascular diseases in adults and children.
CRP, also known as pentraxin 1 (PTX1), is a member of the pentraxin family which has several structurally related members. Most CRP is produced by hepatocytes in the liver induced principally by the inflammatory cytokine interleukin-6 (IL-6), but also by IL-1β and TNF. CRP binds several targets including phosphocholine, a common constituent of polysaccharide coatings of bacterial pathogens and mammalian cell membranes. This allows CRP to function as an opsonin, facilitating phagocytosis of pathogens and dead or dying cells and trigger the classical complement pathway by activating C1q. CRP binding also can stimulate macrophage tumoricidal activity, and protect against septic shock. Another mechanism by which CRP interacts with the innate immune system is through its interaction with Fcγ receptors (FcγRs) on myeloid cells, B lymphocytes, NK cells, and platelets.
Circulating CRP levels in humans are normally quite low, but they increase several hundred-fold during the acute-phase response. Elevated serum CRP levels have been associated with the presence of cardiovascular disease. Numerous research studies investigating serum CRP and its relationship to other diseases have also been reported. These include hypertension, diabetes, cancer, autoimmune disorders, obesity and metabolic syndrome. Additional literature suggests possible links between oral health and chronic infection, inflammation, oxidative stress and heart disease. Studies have also linked elevated serum CRP levels to oral contraceptive use.
For CRP there is encouraging research that suggests salivary CRP may be more strongly associated with serum CRP than is true for other inflammatory markers. CRP is prohibitively large to pass from the circulation into the salivary glands by passive diffusion, and it is believed that small amounts of CRP enter whole saliva mainly as a component of the gingival crevicular fluid (GCF) or through salivary glands. Whole saliva is much easier to collect than GCF, which could make saliva the oral fluid of choice for routine assessment of CRP in clinical or field studies.
As with measurements of salivary cytokines, however, the question of local CRP production in the oral cavity must be considered, since it may obscure the relationship between blood and salivary levels and the interpretation of salivary CRP in relation to other analytes of interest. Although the majority of CRP is thought to originate from the liver, CRP and IL-6 mRNAs have been detected in gingival tissue samples from periodontitis patients and CRP gene expression has been correlated with IL-6 expression.
Several studies have examined the relationship between salivary and serum CRP. One study reported a moderate to strong association between CRP measured in saliva and in serum, while a second longitudinal study found that salivary and plasma CRP were moderately associated cross-sectionally and across two years.
Salivary C-Reactive Protein Assay Principle
This is an indirect sandwich ELISA kit. A “sandwich” is formed when the pre-coated capture anti-CRP antibody present on the plate binds CRP in standards & samples, which is then bound by the anti-CRP detection antibody linked to horseradish peroxidase. After each incubation, unbound components are washed away. Bound anti-CRP Antibody Enzyme Conjugate is then added and the levels measured by the reaction of the horseradish peroxidase (HRP) enzyme to the substrate tetramethylbenzidine (TMB). This reaction produces a blue color. A yellow color is formed after stopping the reaction with an acidic solution. The optical density is read on a standard plate reader at 450 nm. The amount of CRP Antibody Enzyme Conjugate detected is directly proportional to the amount of CRP present in the sample.
3. Technical Summary
| Analyte Summary: | |
|---|---|
| Analyte: | C-Reactive Protein |
| Aliases: | CRP |
| Serum-Saliva Correlation: | NA |
| *Optimum Collection Volume: | 225 μL |
| Special Considerations |
|---|
| Passive Drool is a Preferred Collection Method CRP is Location Dependent |
| Assay Summary: | |
|---|---|
| Methodology: | ELISA |
| Sensitivity: | 1.79 pg/mL |
| Assay Range: | 25 pg/mL - 1600 pg/mL |
| Assay Type: | Quantitative |
Background
C-Reactive Protein (CRP) circulates in the blood plasma as a stable homopentamer and a prominent member of the acute-phase inflammatory proteins. CRP functions mainly in innate immune defense and levels increase in response to inflammation, infection, tissue damage, necrosis, malignancy and allergic reactions (1). CRP has diagnostic value as a marker of systemic inflammation in the body and as an independent risk factor for cardiovascular diseases in adults and children (2, 3). CRP, also known as pentraxin 1 (PTX1), is a member of the pentraxin family which has several structurally related members. Most CRP is produced by hepatocytes in the liver induced principally by the inflammatory cytokine interleukin-6 (IL-6), but also by IL-1β and TNF (4). CRP binds several targets including phosphocholine, a common constituent of polysaccharide coatings of bacterial pathogens and mammalian cell membranes. This allows CRP to function as an opsonin, facilitating phagocytosis of pathogens and dead or dying cells (4, 5) and trigger the classical complement pathway by activating C1q. CRP binding also can stimulate macrophage tumoricidal activity, and protect against septic shock (5). Another mechanism by which CRP interacts with the innate immune system is through its interaction with Fcγ receptors (FcγRs) on myeloid cells, B lymphocytes, NK cells, and platelets (6). Circulating CRP levels in humans are normally quite low, but they increase several hundred-fold during the acute-phase response. Elevated serum CRP levels have been associated with the presence of cardiovascular disease (7, 8). Numerous research studies investigating serum CRP and its relationship to other diseases have also been reported. These include hypertension, (9, 10) diabetes, (11, 12) cancer, (13) autoimmune disorders (14), obesity and metabolic syndrome (15, 16). Additional literature suggests possible links between oral health and chronic infection, inflammation, oxidative stress and heart disease (17),(18). Studies have also linked elevated serum CRP levels to oral contraceptive use (19, 20). For CRP there is encouraging research that suggests salivary CRP may be more strongly associated with serum CRP than is true for other inflammatory markers. CRP is prohibitively large to pass from the circulation into the salivary glands by passive diffusion, and it is believed that small amounts of CRP enter whole saliva mainly as a component of the gingival crevicular fluid (GCF) or through salivary glands. Whole saliva is much easier to collect than GCF, which could make saliva the oral fluid of choice for routine assessment of CRP in clinical or field studies. As with measurements of salivary cytokines, however, the question of local CRP production in the oral cavity must be considered, since it may obscure the relationship between blood and salivary levels and the interpretation of salivary CRP in relation to other analytes of interest. Although the majority of CRP is thought to originate from the liver, CRP and IL-6 mRNAs have been detected in gingival tissue samples from periodontitis patients and CRP gene expression has been correlated with IL-6 expression (21). Several studies have examined the relationship between salivary and serum CRP. One study reported a moderate to strong association between CRP measured in saliva and in serum, while a second longitudinal study found that salivary and plasma CRP were moderately associated cross-sectionally and across two years (22, 23).
References & Salivary C-Reactive Protein Research
-
- Ebersole JL, Cappelli D. Acute-phase reactants in infections and inflammatory diseases. Periodontol 2000. 2000;23:19-49.
- Cook DG, Mendall MA, Whincup PH, Carey IM, Ballam L, Morris JE, et al. C-reactive protein concentration in children: relationship to adiposity and other cardiovascular risk factors. Atherosclerosis. 2000;149(1):139-50.
- Ridker PM, Buring JE, Cook NR, Rifai N. C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: an 8-year follow-up of 14 719 initially healthy American women. Circulation. 2003;107(3):391-7.
- Volanakis JE. Human C-reactive protein: expression, structure, and function. Mol Immunol. 2001;38(2-3):189-97.
- Mortensen RF, Zhong W. Regulation of phagocytic leukocyte activities by C-reactive protein. J Leukoc Biol. 2000;67(4):495-500.
- Nimmerjahn F, Ravetch JV. FcgammaRs in health and disease. Curr Top Microbiol Immunol. 2011;350:105-25.
- Tracy RP, Lemaitre RN, Psaty BM, Ives DG, Evans RW, Cushman M, et al. Relationship of C-reactive protein to risk of cardiovascular disease in the elderly. Results from the Cardiovascular Health Study and the Rural Health Promotion Project. Arterioscler Thromb Vasc Biol. 1997;17(6):1121-7.
- Danesh J, Collins R, Appleby P, Peto R. Association of fibrinogen, C-reactive protein, albumin, or leukocyte count with coronary heart disease: meta-analyses of prospective studies. JAMA. 1998;279(18):1477-82.
- Sesso HD, Buring JE, Rifai N, Blake GJ, Gaziano JM, Ridker PM. C-reactive protein and the risk of developing hypertension. JAMA. 2003;290(22):2945-51.
- Blake GJ, Rifai N, Buring JE, Ridker PM. Blood pressure, C-reactive protein, and risk of future cardiovascular events. Circulation. 2003;108(24):2993-9.
-
- Pradhan AD, Manson JE, Rifai N, Buring JE, Ridker PM. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA. 2001;286(3):327-34.
- Dehghan A, Kardys I, de Maat MP, Uitterlinden AG, Sijbrands EJ, Bootsma AH, et al. Genetic variation, C-reactive protein levels, and incidence of diabetes. Diabetes. 2007;56(3):872-8.
- Erlinger TP, Platz EA, Rifai N, Helzlsouer KJ. C-reactive protein and the risk of incident colorectal cancer. JAMA. 2004;291(5):585-90.
- Du Clos TW. C-reactive protein as a regulator of autoimmunity and inflammation. Arthritis Rheum. 2003;48(6):1475-7.
- Devaraj S, Siegel D, Jialal I. Statin therapy in metabolic syndrome and hypertension post-JUPITER: what is the value of CRP? Curr Atheroscler Rep. 2011;13(1):31-42.
- Luna RC, do Nascimento CC, Asciutti LS, Franceschini Sdo C, Filizola RG, Diniz Ada S, et al. Relation between glucose levels, high-sensitivity C-reactive protein (hs-CRP), body mass index (BMI) and serum and dietary retinol in elderly in population-based study. Arch Gerontol Geriatr. 2012;54(3):462-8.
- Nam K, Jeon Y, Kim TK, Kim KB, Hwang HY, Cho YJ. Intraoperative transfusion and an increased preoperative C-reactive protein level are associated with higher mortality after off-pump coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2019.
- Tonetti, M.S., D’Aiuto, F., Nibali, L., et al. (2007). Treatment of periodontitis and endothelial function. N Engl J Med, 356(9), 911-20.
- Raitakari M, Mansikkaniemi K, Marniemi J, Viikari JS, Raitakari OT. Distribution and determinants of serum high-sensitive C-reactive protein in a population of young adults: The Cardiovascular Risk in Young Finns Study. J Intern Med. 2005;258(5):428-34.
- van Rooijen M, Hansson LO, Frostegard J, Silveira A, Hamsten A, Bremme K. Treatment with combined oral contraceptives induces a rise in serum C-reactive protein in the absence of a general inflammatory response. J Thromb Haemost. 2006;4(1):77-82.
- Lu Q, Jin L. Human gingiva is another site of C-reactive protein formation. J Clin Periodontol. 2010;37(9):789-96.
- Ouellet-Morin I, Danese A, Williams B, Arseneault L. Validation of a high-sensitivity assay for C-reactive protein in human saliva. Brain, behavior, and immunity. 2011;25(4):640-6.
- Out D, Hall RJ, Granger DA, Page GG, Woods SJ. Assessing salivary C-reactive protein: longitudinal associations with systemic inflammation and cardiovascular disease risk in women exposed to intimate partner violence. Brain, behavior, and immunity. 2012;26(4):543-51.
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)