Need Help?
Ask an expert
1. How to collect Salivary Androstenedione
APPROVED SALIVARY ANDROSTENEDIONE COLLECTION METHODS
Salivary Androstenedione Collection Protocol
Collection volume, general considerations, and basic guidelines to maximize salivary androstenedione sample integrity. Use this analyte-specific collection protocol to plan you collection methodology and sampling schemes.

2. How to Assay for Salivary Androstenedione
Send Saliva Samples to Salimetrics
Add to StudyEasy and accurate results from the Salimetrics CoreLab+ Laboratory.
Order Code5149
Salivary Androstenedione ELISA Kit
Add to Study
Salimetrics Assay #1-2902
The Salimetrics Salivary Androstenedione Enzyme Immunoassay Kit is specifically designed to standardize the detection of androstenedione in saliva samples for research and biomedical laboratories. To ensure the most accurate results, this salivary immunoassay is designed using a matrix that matches saliva. The level of androstenedione in saliva (pg/mL) is significantly lower than levels in blood (ng/mL). The standard curve range is sensitive enough to capture individual differences in the androstenedione levels expected in saliva. The current protocol uses only 50 μL of saliva per test and our androstendione assay kit requires no separation or extractions. Read More...| Assay Protocol |
|---|
| Rev. 04.19.19
|
| Specifications | |
|---|---|
| Catalog#: | 1-2902 |
| Regulatory Status: | RUO |
| Format: | 96-well plate |
| Assay Time: | ~ 3 hrs |
| Sample Volume/Test: | 50 µL |
| Sensitivity: | 5 pg/mL |
| Assay Range: | 10 pg/mL - 2430 pg/mL |
| Storage Requirements: | 2-8°C |
| Tests Per Kit | |
|---|---|
| Singlet: | 76 |
| Duplicate: | 38 |
| Target Analyte |
|---|
Technical Documentation
Assay Kit Overview
Intended Use
The Salimetrics Androstenedione Enzyme Immunoassay Kit is a competitive immunoassay specifically designed and validated for the quantitative measurement of salivary Androstenedione. It is not intended for diagnostic use. It is intended only for research use in humans and some animals. Salimetrics has not validated this kit for serum or plasma samples.
Introduction
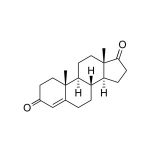
Androstenedione (4-androstene-3,17-dione) is produced in the adrenal gland and gonads. Androstenedione has weak intrinsic androgenic activity (estimated at less than 20% of testosterone), but it is a prohormone for potent androgens (testosterone) and estrogens. The biochemical evidence supporting the effect of Androstenedione on elevation of circulating levels of testosterone and estrogens is strong, and site-of-action direct conversion of Androstenedione to testosterone is well known. Secretion of Androstenedione in women exceeds that of testosterone as significant extra-adrenal conversion of Androstenedione to testosterone occurs in females. High levels of Androstenedione may confer androgenic risk, especially in females, and estrogenic risks, especially in males. Children and adolescents are particularly vulnerable to the effects of Androstenedione conversion to active sex steroids. These effects may disrupt normal sexual development, specifically virilization in girls associated with severe acne, excessive body hair, disruption of the menstrual cycle, and infertility. The conversion of Androstenedione to estrogens can cause feminization of boys. In both boys and girls, the combined effects of excessive androgens and estrogens can induce premature puberty and significantly compromise adult stature by causing early closure of growth plates of long bones. Measurement of serum Androstenedione is used as a marker of androgen biosynthesis. High circulating Androstenedione levels are indicated in virilizing congential adrenal hyperplasia, polycystic ovarian syndrome, and other causes of hirsutism in women. Elevated Androstenedione levels may also occur as a result of adrenal or ovarian tumors. The high serum-saliva correlation for Androstenedione suggests that individual differences in serum Androstenedione levels may be accurately estimated using saliva as a non-invasive alternative specime. The U.S. FDA has released high profile warnings to the public about serious health effects related to Androstenedione administration. Androstenedione is an additive in many products generally advertised as dietary supplements that enhance athletic performance. Use of these supplements has the potential to cause extreme values in this assay. Also, it is noteworthy that the effects of oral Androstenedione administration on circulating levels of testosterone and estrogen do not account for all of the actions of these products. Target tissues of anabolic steroids contain abundant enzymes that convert circulating Androstenedione to testosterone right at the site of action without necessarily affecting circulating testosterone levels. To ensure the most accurate results, this salivary immunoassay is designed using a matrix that matches saliva. The level of Androstenedione in saliva (pg/mL) is significantly lower than levels in the general circulation (ng/mL). The standard curve range of this assay is sensitive enough to capture individual differences in the Androstenedione levels expected in saliva. The current protocol uses only 50 µL of saliva per test. No separation or extractions are necessary.
Androstenedione Assay Principle
This is a competitive immunoassay kit. Androstenedione in standards and samples compete with Androstenedione conjugated to horseradish peroxidase for the antibody binding sites on a microtitre plate. After incubation, unbound components are washed away. Bound Androstenedione Enzyme Conjugate is measured by the reaction of the horseradish peroxidase enzyme to the substrate tetramethylbenzidine (TMB). This reaction produces a blue color. A yellow color is formed after stopping the reaction with an acidic solution. The optical density is read on a standard plate reader at 450 nm. The amount of Androstenedione Enzyme Conjugate detected is inversely proportional to the amount of Androstenedione present in the sample.
3. Technical Summary
| Analyte Summary: | |
|---|---|
| Analyte: | Androstenedione |
| Aliases: | 4-androstenedione, 4-dione; 4-androstene-3,17-dione |
| Serum-Saliva Correlation: | 0.77 |
| *Optimum Collection Volume: | 125 μL |
| Assay Summary: | |
|---|---|
| Methodology: | ELISA |
| Sensitivity: | 5 pg/mL |
| Assay Range: | 10 pg/mL - 2430 pg/mL |
| Assay Type: | Quantitative |
Background
Androstenedione (4-androstenedione; 4-dione; 4-androstene-3,17-dione) is a steroid hormone produced in the adrenal glands and the gonads. (1,2,3) It is synthesized from DHEA or 17OH-progesterone, and it serves principally as the immediate precursor compound that is converted into testosterone or estrone, both of which may then be further converted into estradiol. (4) The same sequence of conversion of DHEA and androstenedione into other steroids also takes place in many peripheral tissues throughout the body, allowing androgens and estrogens to be delivered to the appropriate tissues without leakage of significant amounts into the circulation. (5,6) Levels of androstenedione begin to increase in children at about age 6-8, and it serves as the main source of androgens prior to gonadarche. (7) High levels of androstenedione may confer androgenic risk, especially in females, and estrogenic risks, especially in males. Children and adolescents are particularly vulnerable to the effects of androstenedione conversion to active sex steroids. These effects may disrupt normal sexual development, specifically virilization in girls associated with severe acne, excessive body hair, disruption of the menstrual cycle, and infertility. The conversion of androstenedione to estrogens can cause feminization of boys. (8,9) Elevated levels have been associated with disruptive behavior disorders in children. (7) Measurement of serum androstenedione is used as a marker of androgen biosynthesis. High circulating androstenedione levels are indicated in virilizing congenital adrenal hyperplasia, polycystic ovarian syndrome, and other causes of hirsutism in women. Elevated androstenedione levels may also occur as a result of adrenal or ovarian tumors. (10,11) Androstenedione exhibits a diurnal rhythm similar to that of cortisol, with highest levels in the morning and a nadir in the late evening. (12,13) In blood, androstenedione is not strongly bound to sex hormone binding globulin (SHBG) or albumin. Approximately 95% of circulating androstenedione is available to tissues. (14) Unbound androstenedione enters saliva from blood via intracellular mechanisms, and the correlation between serum and saliva values is highly significant. (15)
References & Salivary Androstenedione Research
-
- Yates, J., Deshpande, N. (1974). Kinetic studies on the enzymes catalyzing the conversion of 17α-hydroxy-progesterone and dehydroepiandrosterone to androstenedione in the human adrenal gland in vitro. J Endocrinol, 6(1), 27-35.
- Carson, R.S., Findlay, J.K., Clarke, I.J., Burger, H.G. (1981). Estradiol, testosterone, and androstenedione in ovine follicular fluid during growth and atresia of ovarian follicles. Biol Reprod, 24(1), 105-113.
- Weinstein, R.L., Kelch, R.P., Jenner, M.R., et al. (1974). Secretion of unconjugated androgens and estrogens by the normal and abnormal human testis before and after human chorionic gonadotropin. J Clin Invest, 53(1), 1-6.
- Dorfman, R. I., Shipley, R. A. (1956). Androgens. New York: John Wiley and Sons.
- Labrie, F., Bélanger, A., Cusan, L., Candas, B. (1997). Physiological changes in dehydroepiandrosterone are not reflected by serum levels of active androgens and estrogens but of their metabolites: Intracrinology. J Clin Endocrinol Metab, 82(8), 2403-9.
- Labrie, F., Luu-The, V. Bélanger, A., et al. (2005). Is dehydroepiandrosterone a hormone? J Endocrinol, 187(2), 169-96.
- Dorn, L.D., Kolko, D.J., Susman, E.J., et al. (2009). Salivary gonadal and adrenal hormone differences in boys and girls with and without disruptive behavior disorders: Contextual variants. Biol Psychol, 81(1), 31-39.
- King, D.S., Sharp, R.L., Vukovich, M.D., et al. (1999). Effect of oral androstenedione on serum testosterone and adaptations to resistance training in young men: A randomized controlled trial. JAMA, 281(21), 2020-8.
- Leder, B.Z., Longcope, C., Catlin, D.H., et al. (2000). Oral androstenedione administration and serum testosterone concentrations in young men. JAMA, 283(6), 779-82.
- Leder, B.Z., Leblanc, K.M., Longcope, C., et al. (2002). Effects of oral androstenedione administration on serum testosterone and estradiol levels in postmenopausal women. J Clin Endocrinol Metab, 87(12), 5449-54.
-
- Kicman, A.T., Bassindale, T., Cowan, D.A., et al. (2003). Effect of androstenedione ingestion on plasma testosterone in young women; A dietary supplement with potential health risks. Clin Chem, 49(1), 167-9.
- Goldman, J., Wajchenberg, B.L., Liberman, B. et al. (1985). Contrast analysis for the evaluation of the circadian rhythms of plasma cortisol, androstenedione, and testosterone in normal men and the possible influence of meals. J Clin Endocrinol Metab, 60(1), 164-67.
- Ostrowska, Z. Zwirska-Korczala, K., Pardela, M., et al. (1998). Circadian variations of androstenedione, dehydroepiandrosterone sulfate and free testosterone in obese women with menstrual disturbances. Endocr Regul, 32(4), 169-76.
- Longcope, C., Baler, S. (1993). Androgen and estrogen dynamics: Relationships with age, weight, and menopause status. J Clin Endocrinol Metab, 76(3), 601–4.
- Vining, R.F., McGinley, R.A. (1987). The measurement of hormones in saliva: Possibilities and pitfalls. J Steroid Biochem, 27(1-3), 81-94.
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)