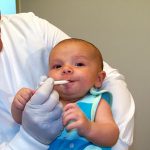
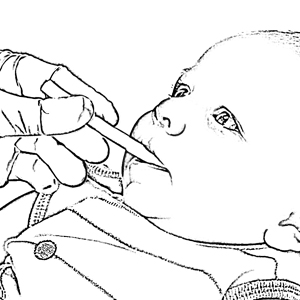
Saliva Collection for Infants <6 Months Old
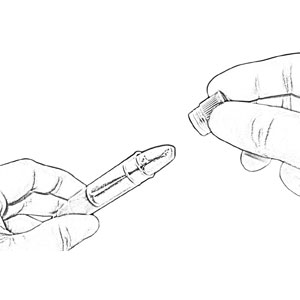
Experience convenient collection from children less than 6 months of age with SalivaBio’s Infant’s Swab (SIS) Participant Sampling Packs. The SIS Method affords you consistent, safe, effective and interference-free swab saliva collection. The SIS method features a 90mm swab which is long enough to hold one end firmly while placing the other end in subject’s mouth (thus eliminating any choking hazard). The SIS’s thin diameter (6.3mm) suits smaller mouths, and the durable polymer withstands chewing. Each swab comes individually wrapped to minimize the possibility of environmental contaminants, causes no change in sample pH, and has verified recoveries. Read More...The volume of sample recovered is typically in the range of 200-1000 µL. The SIS is Also backed by a Salimetrics QC report, which verifies consistent lot-to-lot performance. This method comes complete with SalivaBio Infant’s Swab (SIS), swab storage tube, and sample collection instructions, this saliva collection system is ready to use in the field or on-site.
View the chart below for a list of analytes approved to be collected with the SIS. If your analyte is not listed, we recommend collecting with the passive drool method. However, a pilot study may reveal a high correlation in your subject group, which will enable you to utilize this method effectively.
Technical Summary
| Method Instructions |
|---|
| Specifications | |
|---|---|
| Intended Use: | Infants <6 months old |
| Volume Capacity: | 1 mL |
| Regulatory Status: | FDA Listed, CE Marked |
| Material Composition: | Synthetic Swab |
| Swab Size: | 6.5 mm x 90 mm |
| Individually Wrapped: | Yes |
| Approved for Analytes |
|---|
| Cortisol, Alpha-Amylase, Cotinine, IgG/IgM, Interleukin-1 Beta, Secretory Immunoglobulin A, Testosterone, Uric Acid |
| Technical Documentation |
|---|
| Saliva Collection Handbook
Rigor and Reproducibility: How Good Are Your Saliva Samples? |
How To Collect Saliva: The SalivaBio Infant's Swab
What's Included in Swab Method Sampling Packs?
Pre-Labeled Sampling Packs with Unique Participant ID Scan-Labels
Each ID label is pre-printed with a unique participant ID, study name, and sample number (for individual samples) to simplify organization, scanning, and traceability while reducing errors. Salimetrics labels are made of durable polypropylene to adhere permanently to packs and tubes. Capable of withstanding temperatures below -80°C, and are designed to remain secure through thawing, so no label is lost during sample processing.

Pre-Labeled Sample Swab Storage Tubes (Organized by Study Requirements)
Sample swab storage tubes come pre-labeled with scannable ID labels and are organized specifically for your study design based on your sampling scheme for a streamlined workflow. The Swab Storage Tube has been rigorously validated by Salimetrics to maintain sample integrity for all approved analytes.
| Specifications | |
|---|---|
|
Size: |
17 mm x 100 mm per tube |
| Features: | Can be frozen (to -80° C), swab basket for efficient centrifugation , write on cap |
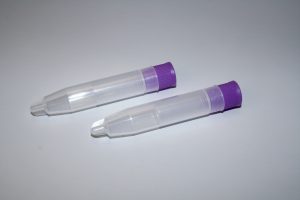
Salimetrics Oral Swabs
The Salimetrics Oral Swab line features swabs for any participant, including animals, to humans of any age group (from infant swabs, to children swabs, to adults swabs, collection of saliva biospecimens from participants is clean, efficient, and features:
- Hygenic, individually wrapped
- FDA Listed, CE Marked
- Simple saliva sample collection
- Validated with saliva/oral fluid specimens & DNA
- Suitable for long-term storage (to -80° C),
- Safe, non-toxic material
- Lot-to-Lot QC for consistent, accurate results
| Specifications | |
|---|---|
|
Size: |
10 x 30 mm (adult), 8 x 125 mm (Children/Animal), 6.5 x 90 mm (Infant) |
| Approved for Biomarkers: | Alpha-Amylase | C-Reactive Protein | Cortisol | Cotinine | IgG/IgM | Interleukin-1 Beta | Interleukin-6 | Osteocalcin | Secretory Immunoglobulin A | Testosterone | DNA Analysis | Uric Acid |
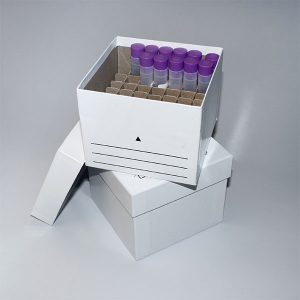
Sample Storage Freezer Boxes
OnTimePoint Saliva Collection Management App (w/CoreLab+ Analysis)
If you are shipping samples back to the Salimetrics CoreLab+, your PSP shipment includes a complimentary study ID for Salimetrics’ OnTimePoint™ (OTP) Saliva Collection Management System. Designed by researchers for researchers, OTP allows you to track the exact time your study samples are collected in real time. This gives you, the researcher, the ability to monitor compliance and take action when needed—ensuring that both participants and their samples remain 100% compliant with saliva collection protocols.
| Features |
|---|
|
CoreLab+ Auto-Roster (w/CoreLab+ Analysis)
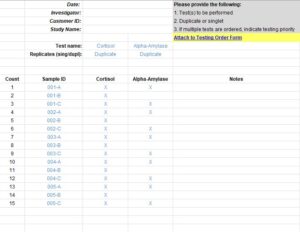
Keep organized in record time. If you’re sending samples to the Salimetrics CoreLab+, Salimetrics will auto-generate and send you a complete XLS sample roster that matches your sample-set for seamless organization. When you’re ready to submit your samples for testing, just specify the biomarkers for each sample you’re sending back to the lab.
Cold-Chain Sample BioShipper (w/CoreLab+ Analysis)
If you are shipping samples back to the Salimetrics CoreLab+, your PSP shipment includes the appropriate quantity and sized bioshipper(s) for returning samples. Bioshippers are required to maintain the cold chain and reliably protect your samples from temperature-related degradation. The CoreLab+ follows best practices for shipping and receiving to ensure your samples remain at the highest possible integrity.
| Specifications | |
|---|---|
|
Size: |
23″ x 16″ x 16″ (large), 14.25″ × 14.25″ × 11.5″ (medium), 16″ x 12″ x 10″ (small) |
| Features: | Bioshipppers are ideal for reliably shipping your saliva samples to Salimetrics CoreLab+ while maintaining the cold-chain to protect your sample integrity. |

How Can we help?
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)
Test Your DBS samples with Salimetrics!
Experts in Saliva & Dried Blood Spot Testing for Research
The best science for your study
OR CALL 800.790.2258
X