(March 6, 2020)
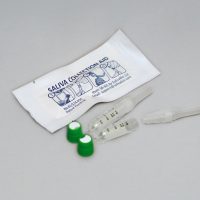
SalivaBio’s Saliva Collection Aid, exclusively from Salimetrics, is the gold standard for saliva collection. The Saliva Collection Aid simplifies the collection of saliva bio-specimens, reduces participant burden, increases compliance and delivers an ideal sample for testing.
The Saliva Collection Aid (SCA), exclusively available through Salimetrics, is a medical device used to facilitate the collection of Passive Drool or whole saliva specimens for storage or laboratory processing. While many researchers choose a collection device without considering the downstream implications to sample quality or processing efficiency, these decisions can be costly to a well-designed study. “Improving the ease and quality of how saliva is collected is often the difference between 90% and 100% compliance,” says Steve Granger, PhD, Salimetrics CSO. “This difference can be even greater depending on the population being tested, such as with the elderly or infirmed patients. That’s why a simple, user-friendly device can have such a big impact.” Best practices in saliva collection methods rely on validated devices and consistent protocols that focus not only on providing accurate results, but also on ensuring all participants, including those who may have difficulty with another saliva collection method, can actually provide the sample needed.
Over the past 20 years, Salimetrics has tested and validated numerous saliva collection devices for salivary bioscience applications. Many early saliva collection techniques in the published literature were found to cause interference with assay quality. “Each year, we repeatedly witness the problems that poorly designed collection devices can cause, and subsequently invest a large amount of time and resources into working with researchers to protect the quality of their data,” says Granger. “Plus, passive drool saliva samples can support a broad range of applications from infectious disease research and DNA analysis, to hormone analysis. Having a tool that maintains sample integrity across an entire range of applications is indispensable.”
Sometimes seeing is believing. “In our Salivary Bioscience Training Workshops, we continue to demonstrate the value that Passive Drool with the Saliva Collection Aid brings to the field,” says Supriya Gaitonde, Ph.D., Salimetrics Senior Applications Scientist. “During the workshops, you can immediately see the positive reaction that researchers have once they’ve seen the Saliva Collection Aid in action. The overall takeaway from 99% of researchers I’ve talked to on the subject agree that using the Saliva Collection Aid would improve sample collection for their participants.”
The Saliva Collection Aid is available to researchers worldwide. Investigators can also experience the simplicity for themselves, by visiting the Salimetrics Website and requesting a Passive Drool Method Sample Pack Online.
About Salimetrics
Salimetrics’ assay kits and CLIA-certified testing services are used to measure salivary analytes related to stress, behavior and development, inflammation, sleep, reproduction, health, and immune function. Founded in 1998 by Douglas A. Granger, Ph.D., Salimetrics, LLC supports CROs, pharmaceuticals, academic researchers and the immunodiagnostic industry around the world with innovative immunoassay products, non-invasive saliva collection methods, and laboratory testing services.
About SalivaBio
SalivaBio is dedicated to the creation of a global standard for collection devices and techniques, as well as the development of new, cutting-edge saliva collection methods to increase ease of use and participant compliance. Exclusively from Salimetrics, all SalivaBio products are extensively tested to minimize or eliminate variability from saliva collection and have been optimized for collection with participant-specific groups.
Read this article on PR Web
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)