Drop Date: January 2020
Latent Trait Cortisol – A Useful Perspective from Your Salivary Bioscience
In This Drop: Latent Trait Cortisol – A Useful Perspective from Your Salivary Bioscience
Salivary Bioscience study designs can take advantage of a participant baseline index for Cortisol available from the study’s saliva samples. In addition to the empirical measurements of Cortisol Awakening Response and Diurnal Slope-Area Under the Curve (AUC), Latent Trait Cortisol can be determined using established statistical modeling to reveal an individual’s trait, or stable cortisol level.
The most empirical saliva sampling indices in Salivary Bioscience by far are the Cortisol Awakening Response (CAR), Diurnal Slope and Area Under the Curve with respect to Ground (AUCg) – all of which require saliva samples to be taken over multiple days and at precise time-points. While saliva sampling indices enable estimation of the trait-like component of variance in biological measures, sources of variability (i.e., measurement error) often compromise the effectiveness of these index-based conclusions. Researchers are constantly struggling with variability due to conflicting evidence to support that saliva sampling is continuously performed in compliance with these study requirements for high-quality data. The majority of this variability is due to sample timing and poor participant compliance where subjects are collecting samples early, late, or not at all. This inconsistent sampling often leads to additional challenges when analyzing salivary cortisol data, but does a better method exist?
Typically, to account for this variability in salivary cortisol indices, researchers use statistical techniques that average samples across datasets. However, this method requires significant statistical power in terms of participants and samples collected to minimize the effects of bias. Based on these challenges, advanced statistical methods, such as Latent Trait Cortisol (LTC), have been developed to account for the shared communalities among multiple saliva samples. A number of studies have explored the LTC model in detail. For example, Doane, et al. (2015), researched the reliability, validity, and stability of LTC levels over several months and reported that “variation in single cortisol measures that were attributable to the corresponding LTC ranged from 20% to 65%.” Doane also showed that “LTC was distinct from the CAR, differentially predicted components of the diurnal profile across the day, and was highly stable across assessment waves (months).”
However, LTC isn’t just statistics, it is also a methodology that can reduce the number of sample collections required for each participant (minimum of 3 samples, Giesbrecht, et al. 2015), diminish participant burden, and increase stability estimates. The result is a latent variable factor that is essentially “free” of state characteristics and measurement error, which increases accuracy beyond that of simply averaging values across samples. This latent variable represents an individual’s trait, or stable, cortisol level.
The main advantage of LTC modeling is that it separates cortisol measurement error into a separate component instead of combining and averaging trait variability with the state variability of cortisol and measurement error. Moreover, even collecting just two cortisol samples per day in multiple waves minimizes cortisol variance and provides a more accurate picture of an individual’s cortisol levels than a single timepoint sample (e.g., Hair cortisol). In addition, single-point measurements deny researchers of any potential for highlighting expected intra-individual cortisol relationships that can’t be examined or identified using single timepoint measures. For example, the LTC modeling approach was implemented in a study by Chen et al, (2017) using the first 2 samples of each day over 3 days. Based on this analysis, LTC modeling enabled researchers to conclude “that serotonergic genetic variation may influence the impact of early adversity on individual differences in HPA-axis regulation.”
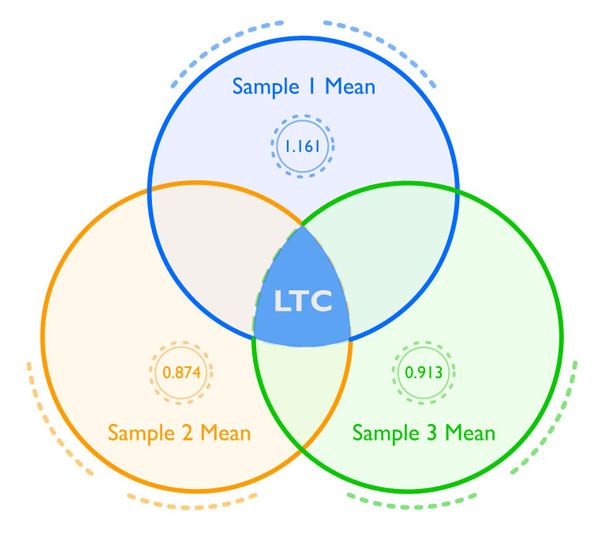
Illustrated in Figure 1 (below) is a way to think about how using a latent modeling statistical approach allows you to find all of the shared information among three samples, or the stable, trait cortisol level. This information is all gathered “behind the scenes” within the statistical program and can be used in this way for any analyses. It’s important to note that the most basic requirements to calculate LTC is a minimum of 3 samples per participant with 30 participants. However, the more participants you have in your study the more robust your results. In addition, it is possible to extricate a LTC score for each person to use in statistical analyses, an approach that is ideal when you have a smaller number of participants.
Overall, researchers continue to advance our understanding of health and human development by incorporating LTC modeling in their salivary cortisol indices. While variability inevitably still exists, understanding and incorporating advanced statistical models can support your key salivary bioscience discovery. If you’re considering LTC modeling in your study, but need more information, feel free to contact the team at Salimetrics and we can help you drive research forward.
REFERENCES & RELATED RESEARCH
- Doane, LD., et al. (2015). Latent trait cortisol (LTC) levels: reliability, validity, and stability. Psychoneuroendocrinology. 55:21-35.
- Chen, FR., et al., (2017). Individual differences in early adolescents’ latent trait cortisol: Interaction of early adversity and 5-HTTLPR. Biol Psychol. 129:8-15.
- Geisbrecht, G., et al. (2015). Latent trait cortisol (LTC) during pregnancy: Composition, continuity, change, and concomitants. Psychoneuroendocrinology. 62:149-58.
- Stroud, CB., et al. (2016). Individual differences in early adolescents’ latent trait cortisol (LTC): Relation to recent acute and chronic stress. Psychoneuroendocrinology. 70:38-46.
*Note: Salimetrics provides this information for research use only (RUO). Information is not provided to promote off-label use of medical devices. Please consult the full-text article.
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)