Description
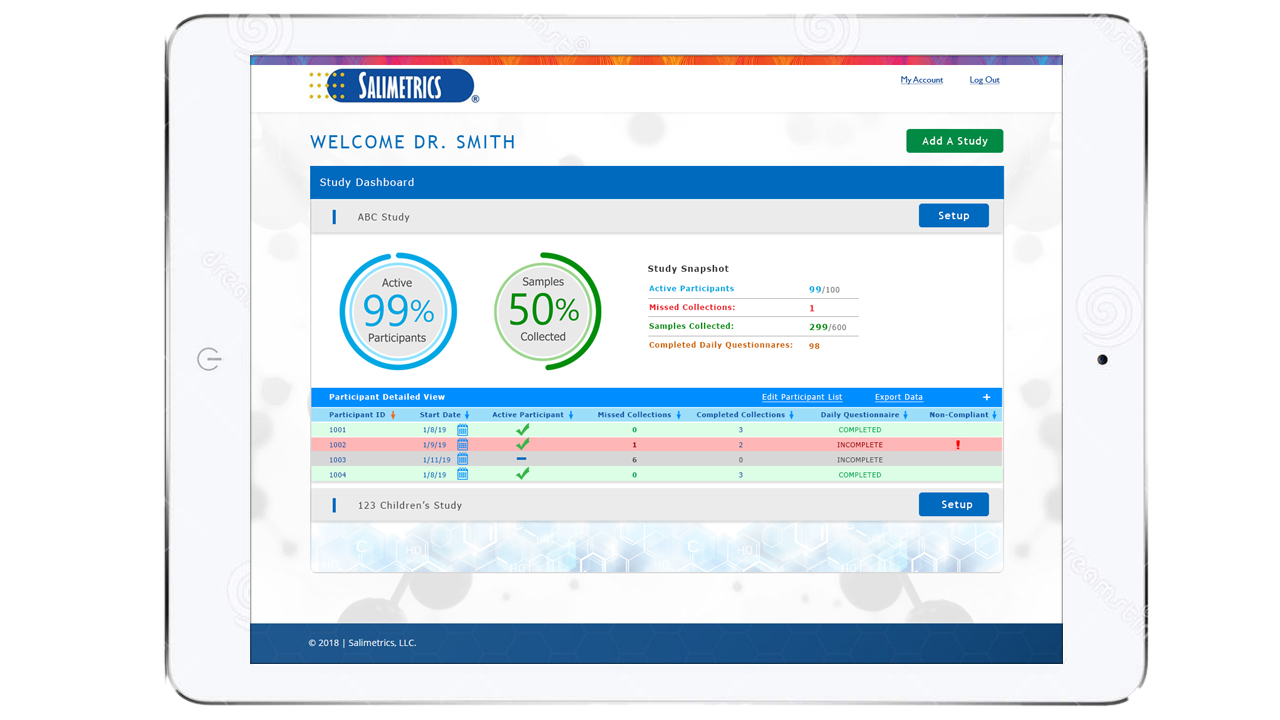
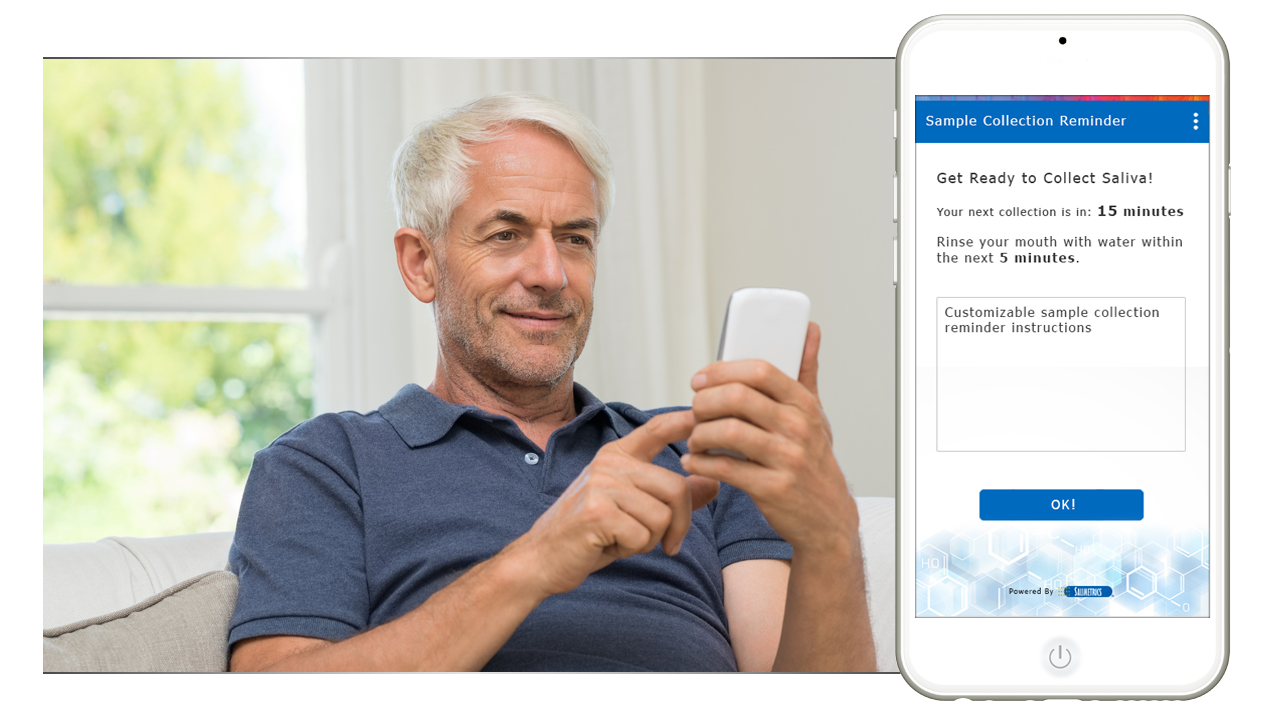
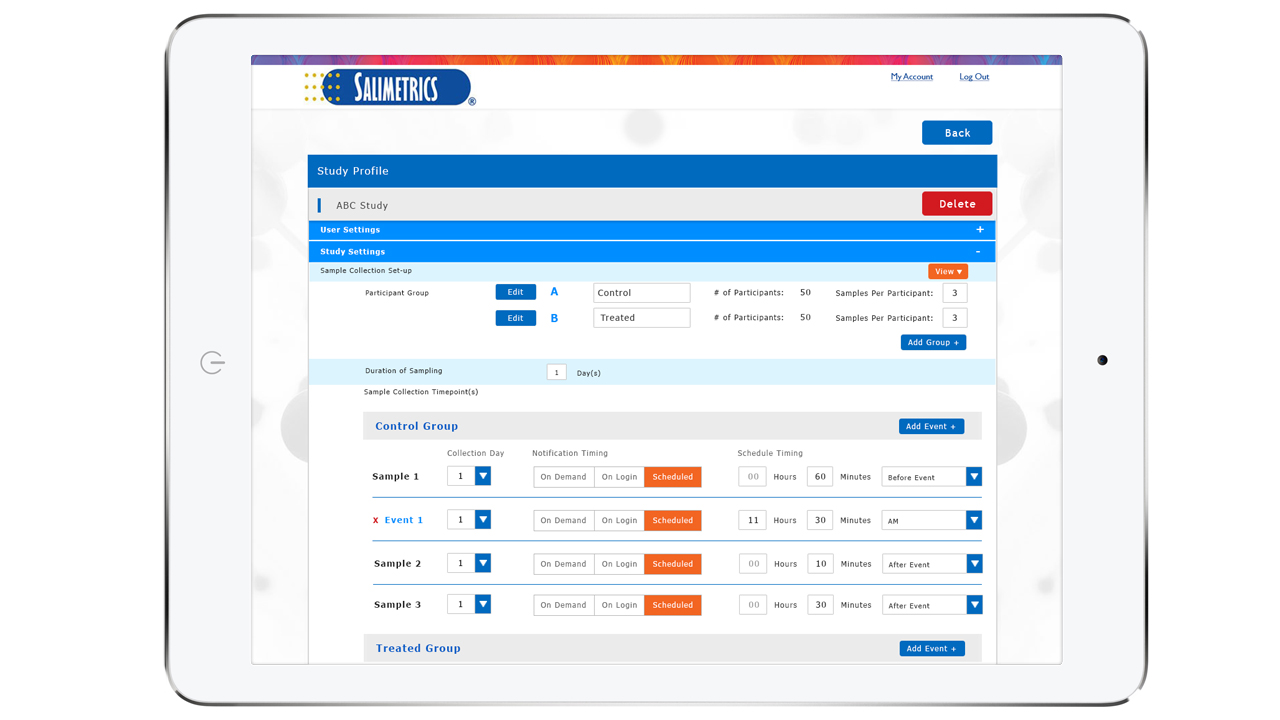
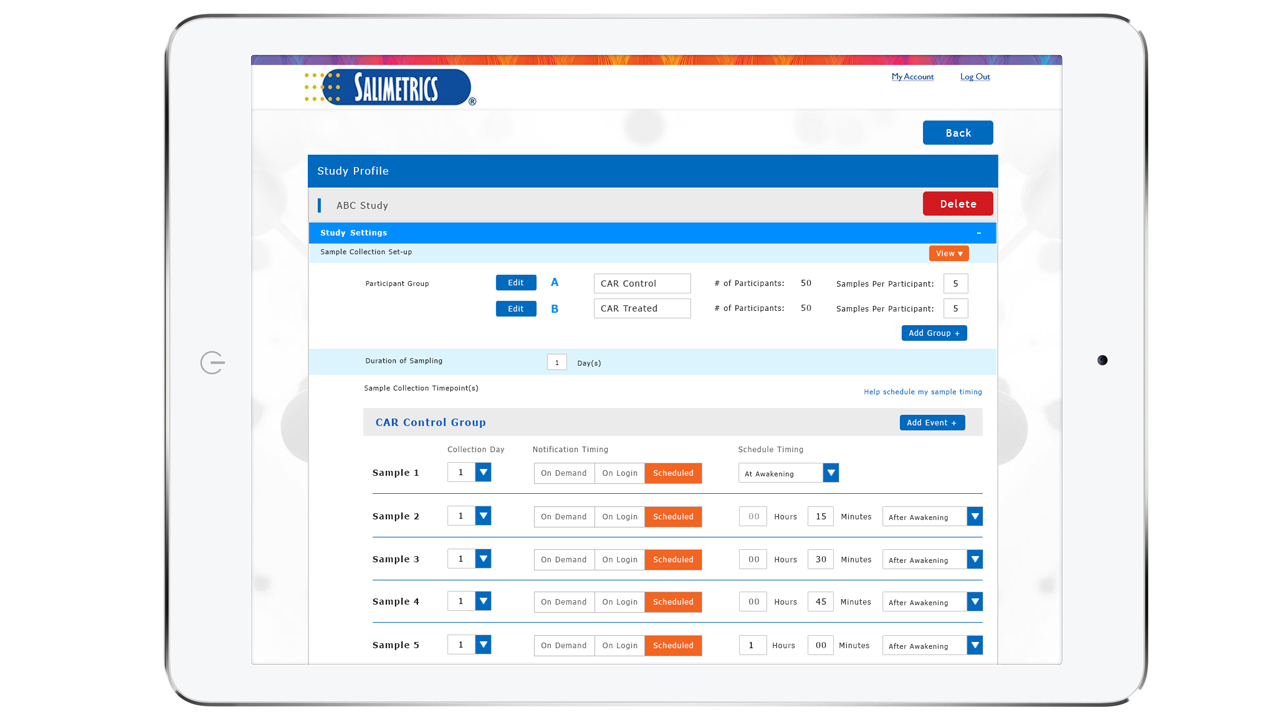
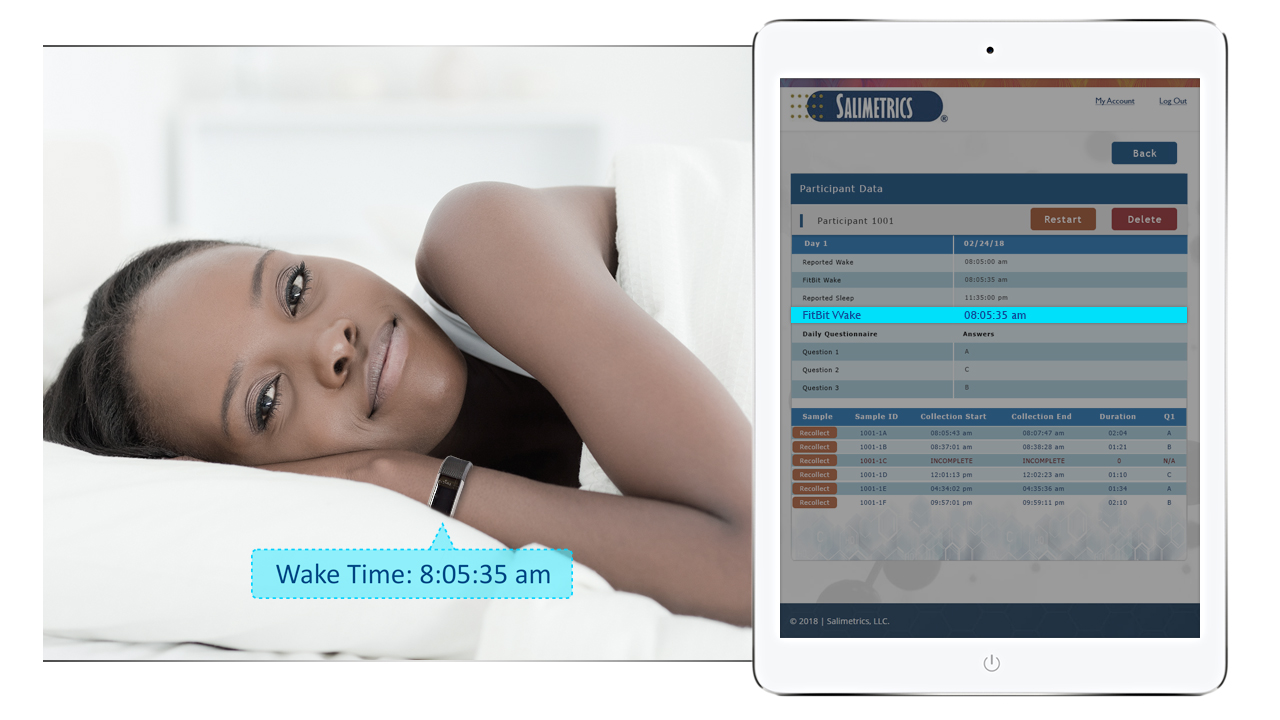
Guarantee time point accuracy with Salimetrics OnTimePoint™ Sample Collection Management System, the only app developed to increase participant compliance and manage sample collection timepoints to ensure researchers and clinicians have the better methods that lead to increased rigor and reproducibility in their Salivary Bioscience. OnTimePoint™ was designed with the features researchers needed to bridge the “salivary bioscience needs gap” – a practical, inexpensive, easy to use, easy to coordinate, easy to implement, easy to monitor Saliva Collection Management System that leverages the better methods of Salivary Bioscience. See the Press Release.
Key Features
- Automated “push” notifications keep participant saliva sample collections on-time
- Actively engage participants during sample collection for real-time validation
- Track up-to-date progress of your complete saliva collection study
- Integrate with wearable devices (FitBit) to know the true diurnal time measurements in addition to self-reported times
- Save >50% of your sample coordinator’s time and budget
See more
 Contact: Salimetrics (USA)
Contact: Salimetrics (USA)